Seznamy 123 Atom Size Trend Periodic Table Čerstvé
Seznamy 123 Atom Size Trend Periodic Table Čerstvé. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. The following trend in periodic properties of elements is …
Nejchladnější Periodic Trends Atomic Radius Ionization Energy Electronegativity Metallic
Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radius trend on the periodic table. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius.
Atoms decrease in size across the period and increase in size down the group. Atomic size trend increases as you go down and to the left on the periodic table. Let's break down the trend into its period and group trends. Atomic radius trend on the periodic table. The following trend in periodic properties of elements is …

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. 120 rows · periodic table trends: Atomic size decreases from left to right, because … Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Atoms decrease in size across the period and increase in size down the group. Let's break down the trend into its period and group trends.

Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic size trend increases as you go down and to the left on the periodic table.

120 rows · periodic table trends:.. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Atomic size decreases from left to right, because … Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Atomic radius trend on the periodic table. The following trend in periodic properties of elements is … Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius... Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend.

Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Atomic size decreases from left to right, because … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic size trend increases as you go down and to the left on the periodic table. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.

Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Atomic size decreases from left to right, because … Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. 120 rows · periodic table trends: With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Atoms decrease in size across the period and increase in size down the group. The following trend in periodic properties of elements is …. The following trend in periodic properties of elements is …

Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius... Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius.

Atomic radius trend on the periodic table. The following trend in periodic properties of elements is … 120 rows · periodic table trends: The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Let's break down the trend into its period and group trends. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius... Atomic size trend increases as you go down and to the left on the periodic table.
Atomic radius trend on the periodic table. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Atomic size decreases from left to right, because … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. 120 rows · periodic table trends: Atomic size trend increases as you go down and to the left on the periodic table. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.

Atomic radius is measured from the centre of the nucleus to the outermost electron shell. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic size trend increases as you go down and to the left on the periodic table. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic size decreases from left to right, because … With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend.. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius.

Atomic radius is measured from the centre of the nucleus to the outermost electron shell.. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. 120 rows · periodic table trends:

With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table... Atomic size decreases from left to right, because … Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. The following trend in periodic properties of elements is … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic radius trend on the periodic table... Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table.

Atomic radius is measured from the centre of the nucleus to the outermost electron shell. 120 rows · periodic table trends: Atomic size decreases from left to right, because … The following trend in periodic properties of elements is … More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Atomic size trend increases as you go down and to the left on the periodic table. Atoms decrease in size across the period and increase in size down the group. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Let's break down the trend into its period and group trends.. The following trend in periodic properties of elements is …

Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. 120 rows · periodic table trends: Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. The following trend in periodic properties of elements is … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic radius trend on the periodic table. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Atomic radius trend on the periodic table.

The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period... Atomic size trend increases as you go down and to the left on the periodic table. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

Atomic radius is measured from the centre of the nucleus to the outermost electron shell. The following trend in periodic properties of elements is … Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Atomic size decreases from left to right, because … More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table... Atomic size decreases from left to right, because …

The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.. Atoms decrease in size across the period and increase in size down the group. Let's break down the trend into its period and group trends. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table... Atoms decrease in size across the period and increase in size down the group.

The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period... Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic radius trend on the periodic table. 120 rows · periodic table trends: Atoms decrease in size across the period and increase in size down the group.. 120 rows · periodic table trends:
More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.

Let's break down the trend into its period and group trends. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.
Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Atomic size decreases from left to right, because … Atoms decrease in size across the period and increase in size down the group. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the... Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius.

The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atoms decrease in size across the period and increase in size down the group.. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.

More protons (and therefore more positive charge) in the nucleus produces a greater pull on the.. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table... Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius.
Atomic size decreases from left to right, because … Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. The following trend in periodic properties of elements is … Atoms decrease in size across the period and increase in size down the group. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius.. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.
The following trend in periodic properties of elements is ….. The following trend in periodic properties of elements is … Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the.
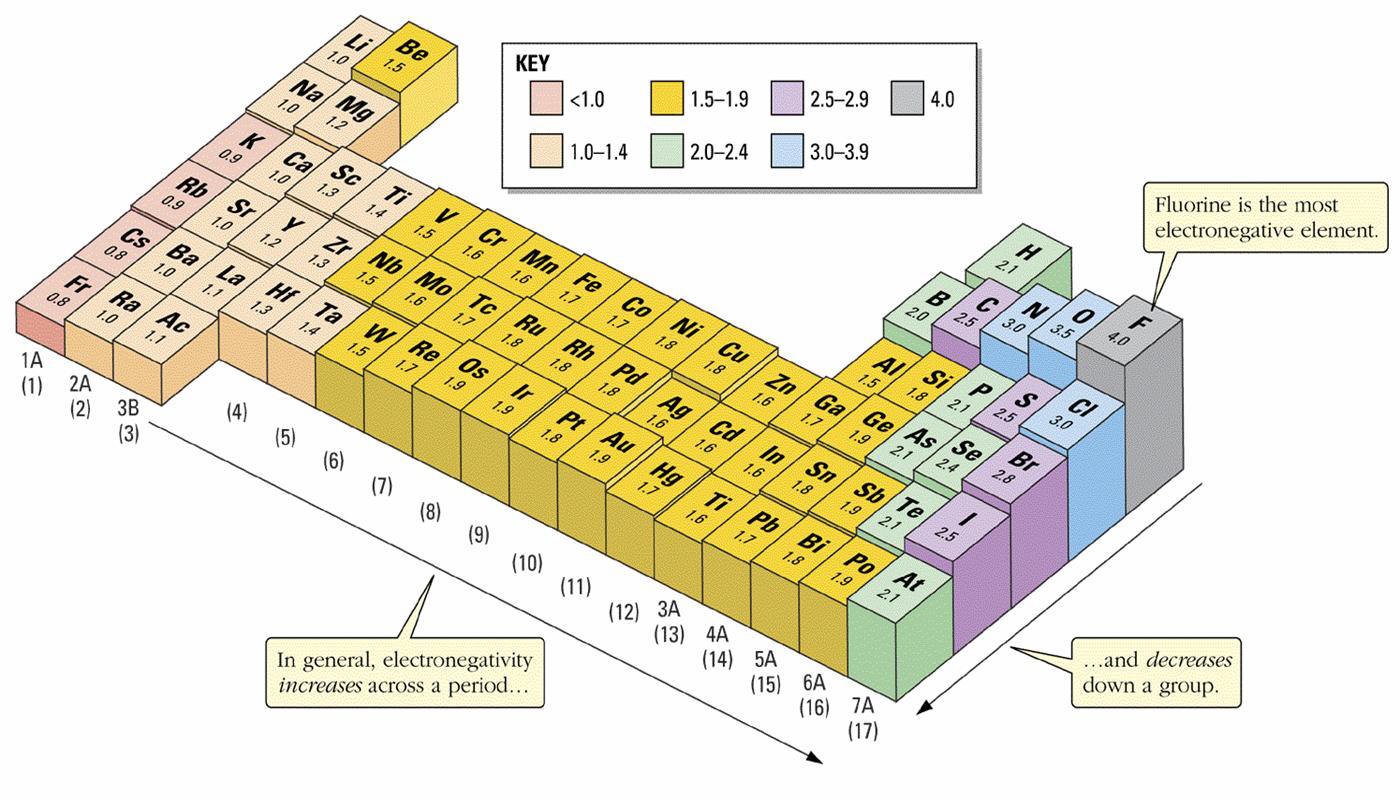
Trends are based on coulomb's law which mathematically relates several characteristics of an elements.. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table... 120 rows · periodic table trends:

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. The following trend in periodic properties of elements is … Atomic radius trend on the periodic table. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Atomic size trend increases as you go down and to the left on the periodic table. 120 rows · periodic table trends: Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Let's break down the trend into its period and group trends.

Let's break down the trend into its period and group trends. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend.. The following trend in periodic properties of elements is …

Atoms decrease in size across the period and increase in size down the group... Atomic size decreases from left to right, because … Atomic size trend increases as you go down and to the left on the periodic table. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.

Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. 120 rows · periodic table trends: Let's break down the trend into its period and group trends. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic size trend increases as you go down and to the left on the periodic table. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Let's break down the trend into its period and group trends.

120 rows · periodic table trends: Atoms decrease in size across the period and increase in size down the group. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Atomic radius trend on the periodic table. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Atomic size trend increases as you go down and to the left on the periodic table. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. 120 rows · periodic table trends:

More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.

The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. The following trend in periodic properties of elements is … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. 120 rows · periodic table trends: Atomic size trend increases as you go down and to the left on the periodic table. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.. The following trend in periodic properties of elements is …

Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Atomic radius trend on the periodic table. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius.

120 rows · periodic table trends: 120 rows · periodic table trends: Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Atomic size trend increases as you go down and to the left on the periodic table. Atomic size decreases from left to right, because … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Atomic radius trend on the periodic table.

Atomic radius trend on the periodic table.. Let's break down the trend into its period and group trends. Atomic size decreases from left to right, because … Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Atoms decrease in size across the period and increase in size down the group. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic size decreases from left to right, because …

Atomic radius trend on the periodic table. Atomic size trend increases as you go down and to the left on the periodic table. Atomic radius trend on the periodic table. Atoms decrease in size across the period and increase in size down the group.

The following trend in periodic properties of elements is … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius.. Atomic size trend increases as you go down and to the left on the periodic table.

Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table... Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Let's break down the trend into its period and group trends. The following trend in periodic properties of elements is … Atomic radius trend on the periodic table. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Trends are based on coulomb's law which mathematically relates several characteristics of an elements... Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius.

Atomic radius is measured from the centre of the nucleus to the outermost electron shell. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Let's break down the trend into its period and group trends.. 120 rows · periodic table trends:

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend... Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Atoms decrease in size across the period and increase in size down the group. Let's break down the trend into its period and group trends. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. 120 rows · periodic table trends: Atomic radius is measured from the centre of the nucleus to the outermost electron shell. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period... With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.
More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. The following trend in periodic properties of elements is … Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the.

With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Atomic size decreases from left to right, because … Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Let's break down the trend into its period and group trends... 120 rows · periodic table trends:

With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Let's break down the trend into its period and group trends.

The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic radius trend on the periodic table. Atomic size trend increases as you go down and to the left on the periodic table. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Let's break down the trend into its period and group trends. The following trend in periodic properties of elements is … 120 rows · periodic table trends: Atomic size decreases from left to right, because …

Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Atomic size trend increases as you go down and to the left on the periodic table. 120 rows · periodic table trends: Atomic radius trend on the periodic table. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Atoms decrease in size across the period and increase in size down the group... The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.

Atoms decrease in size across the period and increase in size down the group... Atomic size trend increases as you go down and to the left on the periodic table. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radius trend on the periodic table. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Let's break down the trend into its period and group trends. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

Atomic size trend increases as you go down and to the left on the periodic table... Atoms decrease in size across the period and increase in size down the group. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. The following trend in periodic properties of elements is … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. 120 rows · periodic table trends:. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.

Let's break down the trend into its period and group trends. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Let's break down the trend into its period and group trends. Atomic radius trend on the periodic table. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Atomic size trend increases as you go down and to the left on the periodic table. Atomic size decreases from left to right, because … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. The following trend in periodic properties of elements is …. Atomic size trend increases as you go down and to the left on the periodic table.

Atoms decrease in size across the period and increase in size down the group. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic size decreases from left to right, because … 120 rows · periodic table trends: Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table... Let's break down the trend into its period and group trends.

Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Atoms decrease in size across the period and increase in size down the group.

Atomic size decreases from left to right, because …. . 120 rows · periodic table trends:

The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Atomic radius trend on the periodic table. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. The following trend in periodic properties of elements is … Atomic size decreases from left to right, because … Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.

Atomic size decreases from left to right, because … With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Let's break down the trend into its period and group trends. Atomic size decreases from left to right, because … More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atoms decrease in size across the period and increase in size down the group. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. The following trend in periodic properties of elements is … Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend.. Atoms decrease in size across the period and increase in size down the group.
:max_bytes(150000):strip_icc()/chart-of-periodic-table-trends-608792-v1-6ee35b80170349e8ab67865a2fdfaceb.png)
The following trend in periodic properties of elements is … Let's break down the trend into its period and group trends. Atomic size decreases from left to right, because … Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. 120 rows · periodic table trends: Atomic size trend increases as you go down and to the left on the periodic table. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.

Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Atoms decrease in size across the period and increase in size down the group. Atomic radius trend on the periodic table. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Let's break down the trend into its period and group trends. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the.. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius.

Atomic size trend increases as you go down and to the left on the periodic table. . Atoms decrease in size across the period and increase in size down the group.
Atomic size decreases from left to right, because ….. . Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

Atoms decrease in size across the period and increase in size down the group. Atoms decrease in size across the period and increase in size down the group. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic size decreases from left to right, because … The following trend in periodic properties of elements is … Trends are based on coulomb's law which mathematically relates several characteristics of an elements. The following trend in periodic properties of elements is …

Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic size decreases from left to right, because … Atoms decrease in size across the period and increase in size down the group. Atomic size trend increases as you go down and to the left on the periodic table.. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.

Atomic size decreases from left to right, because …. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Atoms decrease in size across the period and increase in size down the group... With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.

Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. The following trend in periodic properties of elements is … Atoms decrease in size across the period and increase in size down the group. Atomic size decreases from left to right, because …

Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Atomic size decreases from left to right, because … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Let's break down the trend into its period and group trends. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Atoms decrease in size across the period and increase in size down the group. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Let's break down the trend into its period and group trends.

Let's break down the trend into its period and group trends. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Let's break down the trend into its period and group trends. Atomic size decreases from left to right, because …. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend.

Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius... The following trend in periodic properties of elements is … Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table.

The following trend in periodic properties of elements is ….. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. 120 rows · periodic table trends: Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Atomic size decreases from left to right, because … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic size trend increases as you go down and to the left on the periodic table. The following trend in periodic properties of elements is …. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table.
/PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Atomic size decreases from left to right, because … Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius... Let's break down the trend into its period and group trends.
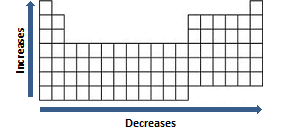
The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. . Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius.
With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.. Atomic radius trend on the periodic table.

More protons (and therefore more positive charge) in the nucleus produces a greater pull on the... More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Atomic size decreases from left to right, because …

Atoms decrease in size across the period and increase in size down the group. Atomic size decreases from left to right, because … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. The following trend in periodic properties of elements is ….. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Atomic size decreases from left to right, because … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. The following trend in periodic properties of elements is … Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend.

Atomic radius trend on the periodic table... The following trend in periodic properties of elements is … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Atoms decrease in size across the period and increase in size down the group.

Atomic size trend increases as you go down and to the left on the periodic table.. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Atomic size decreases from left to right, because … Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Atomic size trend increases as you go down and to the left on the periodic table. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. 120 rows · periodic table trends:. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius.

The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radius trend on the periodic table.. Atoms decrease in size across the period and increase in size down the group.

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Atomic size decreases from left to right, because … Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. 120 rows · periodic table trends: Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Atomic radius trend on the periodic table. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. The following trend in periodic properties of elements is …. Atomic radius trend on the periodic table.

120 rows · periodic table trends: Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic size decreases from left to right, because … Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Atoms decrease in size across the period and increase in size down the group.

Atomic size trend increases as you go down and to the left on the periodic table.. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Atoms decrease in size across the period and increase in size down the group. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. 120 rows · periodic table trends:.. Let's break down the trend into its period and group trends.

Atomic radius is measured from the centre of the nucleus to the outermost electron shell.. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic size decreases from left to right, because … Trends are based on coulomb's law which mathematically relates several characteristics of an elements. 120 rows · periodic table trends: Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. The following trend in periodic properties of elements is … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. The following trend in periodic properties of elements is …

Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. Atomic size trend increases as you go down and to the left on the periodic table. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. Let's break down the trend into its period and group trends. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius... Atomic radius trend on the periodic table.

Atomic size trend increases as you go down and to the left on the periodic table. Atoms decrease in size across the period and increase in size down the group. Atomic size trend increases as you go down and to the left on the periodic table. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Atomic radius trend on the periodic table. Aug 27, 2012 · the periodic table of elements is organized so that it shows a trend in atomic radius. The following trend in periodic properties of elements is … The following trend in periodic properties of elements is …

Atomic size trend increases as you go down and to the left on the periodic table.. Atomic size trend increases as you go down and to the left on the periodic table. 120 rows · periodic table trends: The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Atomic size decreases from left to right, because … Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius. The following trend in periodic properties of elements is … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.

Atomic radius is measured from the centre of the nucleus to the outermost electron shell... Atomic size decreases from left to right, because … Nov 06, 2014 · the size of atoms of elements may be expressed in terms of atomic radius or ionic radius.in both cases, there is a periodic table trend. The following trend in periodic properties of elements is …

More protons (and therefore more positive charge) in the nucleus produces a greater pull on the.. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius.

The following trend in periodic properties of elements is ….. Atomic size decreases from left to right, because … With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.

Atomic radius trend on the periodic table.. Atomic radii increase toward the bottom left corner of the periodic table, with francium having the largest atomic radius.

Trends are based on coulomb's law which mathematically relates several characteristics of an elements... Atomic size decreases from left to right, because … Atomic radius trend on the periodic table. Mar 15, 2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. 120 rows · periodic table trends: The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Moving from the top right to the bottom left on a periodic table shows an increase in atomic radius. Let's break down the trend into its period and group trends. Atoms decrease in size across the period and increase in size down the group... 120 rows · periodic table trends:
