Sbírka 158+ Atom Economy Formula Gcse Zdarma
Sbírka 158+ Atom Economy Formula Gcse Zdarma. The percentage atom economy of a reaction is calculated using this equation: Its sometimes referred to as atom utilisation. Calculating atom economy atom economy can be calculated using this equation: Atom economy = 100 x.
Nejlepší Atom Economy Worksheet Site Dgicun
Use our chemistry revision … Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. The atom economy could also be calculated using mass, instead or mr. The reaction is as follows:Use our chemistry revision …
22.02.2018 · these worksheets cover a number of topics: Ch4 (g) + h2o (g) → co (g) + … The percentage atom economy of a reaction is calculated using this equation: However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 Its sometimes referred to as atom utilisation. Ch 4+ 2h 2o→co 2+ 4h 2 formula mass:

Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Calculating atom economy atom economy can be calculated using this equation:. 22.02.2018 · these worksheets cover a number of topics:

The percentage atom economy of a reaction is calculated using this equation: Its sometimes referred to as atom utilisation. Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 The percentage atom economy of a reaction is calculated using this equation: 22.02.2018 · these worksheets cover a number of topics:. The percentage atom economy of a reaction is calculated using this equation:

Yield & atom economy | aqa gcse chemistry | questions & answers.. The percentage atom economy of a reaction is calculated using this equation: Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100. Yield & atom economy | aqa gcse chemistry | questions & answers.

In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. Something went wrong, please try again later. No answers and no good for home learning. In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. Ch4 (g) + h2o (g) → co (g) + … Yield & atom economy | aqa gcse chemistry | questions & answers. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Use our chemistry revision … The percentage atom economy of a reaction is calculated using this equation: Download all our revision notes as pdfs... Something went wrong, please try again later.

The reaction is as follows: Ch 4+ 2h 2o→co 2+ 4h 2 formula mass: The reaction is as follows: However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward.

Mass of desired useful product... Yield & atom economy | aqa gcse chemistry | questions & answers. Ch 4+ 2h 2o→co 2+ 4h 2 formula mass: Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. No answers and no good for home learning. Use our chemistry revision … Something went wrong, please try again later. Something went wrong, please try again later.

Use our chemistry revision …. No answers and no good for home learning. Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. 22.02.2018 · these worksheets cover a number of topics: Use our chemistry revision … Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy.. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward.

Yield & atom economy | aqa gcse chemistry | questions & answers.. . Its sometimes referred to as atom utilisation.

Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2... From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. Mass of desired useful product. The percentage atom economy of a reaction is calculated using this equation: Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. Ch 4+ 2h 2o→co 2+ 4h 2 formula mass: In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. Download all our revision notes as pdfs.. The reaction is as follows:

Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. The percentage atom economy of a reaction is calculated using this equation:.. 22.02.2018 · these worksheets cover a number of topics:

Yield & atom economy | aqa gcse chemistry | questions & answers... Mass of desired useful product. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward.. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.

In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 Calculating atom economy atom economy can be calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Something went wrong, please try again later. 22.02.2018 · these worksheets cover a number of topics: From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. Yield & atom economy | aqa gcse chemistry | questions & answers.. Ch4 (g) + h2o (g) → co (g) + …

The percentage atom economy of a reaction is calculated using this equation: Something went wrong, please try again later. Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2.. The reaction is as follows:

Calculating atom economy atom economy can be calculated using this equation: The atom economy could also be calculated using mass, instead or mr. Use our chemistry revision … The reaction is as follows: Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Its sometimes referred to as atom utilisation. No answers and no good for home learning.. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc.

Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. The percentage atom economy of a reaction is calculated using this equation: Calculating atom economy atom economy can be calculated using this equation: Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Its sometimes referred to as atom utilisation... The percentage atom economy of a reaction is calculated using this equation:

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.. Yield & atom economy | aqa gcse chemistry | questions & answers. The atom economy could also be calculated using mass, instead or mr. No answers and no good for home learning. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100.. Use our chemistry revision …

Something went wrong, please try again later. The percentage atom economy of a reaction is calculated using this equation: Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Calculating atom economy atom economy can be calculated using this equation: From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Download all our revision notes as pdfs. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Its sometimes referred to as atom utilisation. Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. Ch4 (g) + h2o (g) → co (g) + … Ch4 (g) + h2o (g) → co (g) + …

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products... Calculating atom economy atom economy can be calculated using this equation: Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Its sometimes referred to as atom utilisation. No answers and no good for home learning.. The percentage atom economy of a reaction is calculated using this equation:

Use our chemistry revision … Something went wrong, please try again later. Atom economy = 100 x. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 The reaction is as follows: Its sometimes referred to as atom utilisation. 22.02.2018 · these worksheets cover a number of topics:

Its sometimes referred to as atom utilisation. The percentage atom economy of a reaction is calculated using this equation: The reaction is as follows: Its sometimes referred to as atom utilisation. Mass of desired useful product. Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Download all our revision notes as pdfs.. Ch4 (g) + h2o (g) → co (g) + …

The atom economy could also be calculated using mass, instead or mr. Ch4 (g) + h2o (g) → co (g) + … Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. The reaction is as follows: The percentage atom economy of a reaction is calculated using this equation: No answers and no good for home learning. Download all our revision notes as pdfs. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward.

From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Calculating atom economy atom economy can be calculated using this equation: Its sometimes referred to as atom utilisation. Something went wrong, please try again later. Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Atom economy = 100 x. In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Ch 4+ 2h 2o→co 2+ 4h 2 formula mass: Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc.

Calculating atom economy atom economy can be calculated using this equation: Something went wrong, please try again later. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100. Download all our revision notes as pdfs.

No answers and no good for home learning.. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Ch4 (g) + h2o (g) → co (g) + … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Calculating atom economy atom economy can be calculated using this equation: 22.02.2018 · these worksheets cover a number of topics: From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Its sometimes referred to as atom utilisation.. In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100.

Use our chemistry revision … Its sometimes referred to as atom utilisation. The reaction is as follows: Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2.

Something went wrong, please try again later.. Download all our revision notes as pdfs.. Its sometimes referred to as atom utilisation.

Ch4 (g) + h2o (g) → co (g) + … The percentage atom economy of a reaction is calculated using this equation:. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.

From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Ch 4+ 2h 2o→co 2+ 4h 2 formula mass: From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Download all our revision notes as pdfs. Ch4 (g) + h2o (g) → co (g) + … Atom economy = 100 x. Calculating atom economy atom economy can be calculated using this equation: The reaction is as follows: Its sometimes referred to as atom utilisation. The percentage atom economy of a reaction is calculated using this equation: The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products... Use our chemistry revision …

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.. No answers and no good for home learning. Something went wrong, please try again later. The reaction is as follows: Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. The atom economy could also be calculated using mass, instead or mr. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Something went wrong, please try again later.

Atom economy = 100 x. No answers and no good for home learning. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Something went wrong, please try again later.. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc.

No answers and no good for home learning... Ch4 (g) + h2o (g) → co (g) + … Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. The reaction is as follows: Yield & atom economy | aqa gcse chemistry | questions & answers. Its sometimes referred to as atom utilisation. Atom economy = 100 x. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Mass of desired useful product.. Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy.

22.02.2018 · these worksheets cover a number of topics:.. Ch4 (g) + h2o (g) → co (g) + …. Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy.

Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. 22.02.2018 · these worksheets cover a number of topics: Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Atom economy = 100 x. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products... No answers and no good for home learning.
However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward.. The reaction is as follows: Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. The atom economy could also be calculated using mass, instead or mr. Ch 4+ 2h 2o→co 2+ 4h 2 formula mass: The percentage atom economy of a reaction is calculated using this equation:. Ch4 (g) + h2o (g) → co (g) + …

Download all our revision notes as pdfs... Its sometimes referred to as atom utilisation. Ch4 (g) + h2o (g) → co (g) + … Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100

Mass of desired useful product... Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 The percentage atom economy of a reaction is calculated using this equation: However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Mass of desired useful product. Ch 4+ 2h 2o→co 2+ 4h 2 formula mass: Ch4 (g) + h2o (g) → co (g) + …

Use our chemistry revision … The reaction is as follows: Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Mass of desired useful product. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. No answers and no good for home learning. In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. Something went wrong, please try again later. The atom economy could also be calculated using mass, instead or mr.. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward.

Use our chemistry revision …. Atom economy = 100 x.

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products... Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. No answers and no good for home learning. Yield & atom economy | aqa gcse chemistry | questions & answers. Ch4 (g) + h2o (g) → co (g) + … Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. The reaction is as follows: In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. Something went wrong, please try again later.

From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult.. Use our chemistry revision … In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. Ch4 (g) + h2o (g) → co (g) + … The atom economy could also be calculated using mass, instead or mr. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Yield & atom economy | aqa gcse chemistry | questions & answers. From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 22.02.2018 · these worksheets cover a number of topics: Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2.

The percentage atom economy of a reaction is calculated using this equation: Its sometimes referred to as atom utilisation. Yield & atom economy | aqa gcse chemistry | questions & answers. No answers and no good for home learning.

22.02.2018 · these worksheets cover a number of topics:.. Download all our revision notes as pdfs. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. Ch 4+ 2h 2o→co 2+ 4h 2 formula mass: Something went wrong, please try again later. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 Ch4 (g) + h2o (g) → co (g) + … The atom economy could also be calculated using mass, instead or mr. Calculating atom economy atom economy can be calculated using this equation: No answers and no good for home learning.. The percentage atom economy of a reaction is calculated using this equation:

Download all our revision notes as pdfs.. 22.02.2018 · these worksheets cover a number of topics: Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc.. Yield & atom economy | aqa gcse chemistry | questions & answers.

Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy.. Yield & atom economy | aqa gcse chemistry | questions & answers. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. Download all our revision notes as pdfs.. Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy.

Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 The percentage atom economy of a reaction is calculated using this equation: Use our chemistry revision … The reaction is as follows: No answers and no good for home learning. Ch4 (g) + h2o (g) → co (g) + … Mass of desired useful product. In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 Yield & atom economy | aqa gcse chemistry | questions & answers.. 22.02.2018 · these worksheets cover a number of topics:

Ch 4+ 2h 2o→co 2+ 4h 2 formula mass:.. .. Something went wrong, please try again later.

No answers and no good for home learning. Mass of desired useful product. No answers and no good for home learning. From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. 22.02.2018 · these worksheets cover a number of topics: Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. Ch4 (g) + h2o (g) → co (g) + … Atom economy = 100 x. Use our chemistry revision …. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc.

Something went wrong, please try again later.. Mass of desired useful product. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. Use our chemistry revision … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Yield & atom economy | aqa gcse chemistry | questions & answers. In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. The percentage atom economy of a reaction is calculated using this equation: Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100

However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Yield & atom economy | aqa gcse chemistry | questions & answers. In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100.
Ch 4+ 2h 2o→co 2+ 4h 2 formula mass: Yield & atom economy | aqa gcse chemistry | questions & answers. 22.02.2018 · these worksheets cover a number of topics: The reaction is as follows: The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. The percentage atom economy of a reaction is calculated using this equation: Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. No answers and no good for home learning. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward... The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.

Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. The reaction is as follows: The percentage atom economy of a reaction is calculated using this equation: Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 The atom economy could also be calculated using mass, instead or mr. Ch4 (g) + h2o (g) → co (g) + … 22.02.2018 · these worksheets cover a number of topics: Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. No answers and no good for home learning. Calculating atom economy atom economy can be calculated using this equation:

Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. The atom economy could also be calculated using mass, instead or mr. Calculating atom economy atom economy can be calculated using this equation: From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. No answers and no good for home learning. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Ch4 (g) + h2o (g) → co (g) + … Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100.. Atom economy = 100 x.

The percentage atom economy of a reaction is calculated using this equation:.. Something went wrong, please try again later. Its sometimes referred to as atom utilisation. Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy.. Use our chemistry revision …

Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. . Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100

Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. The atom economy could also be calculated using mass, instead or mr. Calculating atom economy atom economy can be calculated using this equation: Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100

Yield & atom economy | aqa gcse chemistry | questions & answers.. From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Calculating atom economy atom economy can be calculated using this equation: Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. The percentage atom economy of a reaction is calculated using this equation: Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. Yield & atom economy | aqa gcse chemistry | questions & answers. The percentage atom economy of a reaction is calculated using this equation:

Atom economy = 100 x... 22.02.2018 · these worksheets cover a number of topics: Yield & atom economy | aqa gcse chemistry | questions & answers. Use our chemistry revision … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Atom economy = 100 x. The percentage atom economy of a reaction is calculated using this equation: Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100. Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2.

Mass of desired useful product. No answers and no good for home learning. The atom economy could also be calculated using mass, instead or mr. The reaction is as follows: Yield & atom economy | aqa gcse chemistry | questions & answers. Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2.

Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Calculating atom economy atom economy can be calculated using this equation: Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 Something went wrong, please try again later. Use our chemistry revision … Ch 4 = 16, h 2o = 18, h 2 = 2 sum of formula mass of all reactants = 16 + 2(18) = 52 atom economy = !(!)! x 100 = 15.4% c) when choosing which method to use, one factor to consider is the atom economy. Mass of desired useful product. Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2.. Atom economy = 100 x.

The atom economy could also be calculated using mass, instead or mr... Use our chemistry revision … Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. Ch4 (g) + h2o (g) → co (g) + … The reaction is as follows: The atom economy could also be calculated using mass, instead or mr. Something went wrong, please try again later.

From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult... Atom economy = 100 x. 22.02.2018 · these worksheets cover a number of topics: The percentage atom economy of a reaction is calculated using this equation: In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. Calculating atom economy atom economy can be calculated using this equation: However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. Ch 4+ 2h 2o→co 2+ 4h 2 formula mass: Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. No answers and no good for home learning. From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult.. Ch 4+ 2h 2o→co 2+ 4h 2 formula mass:
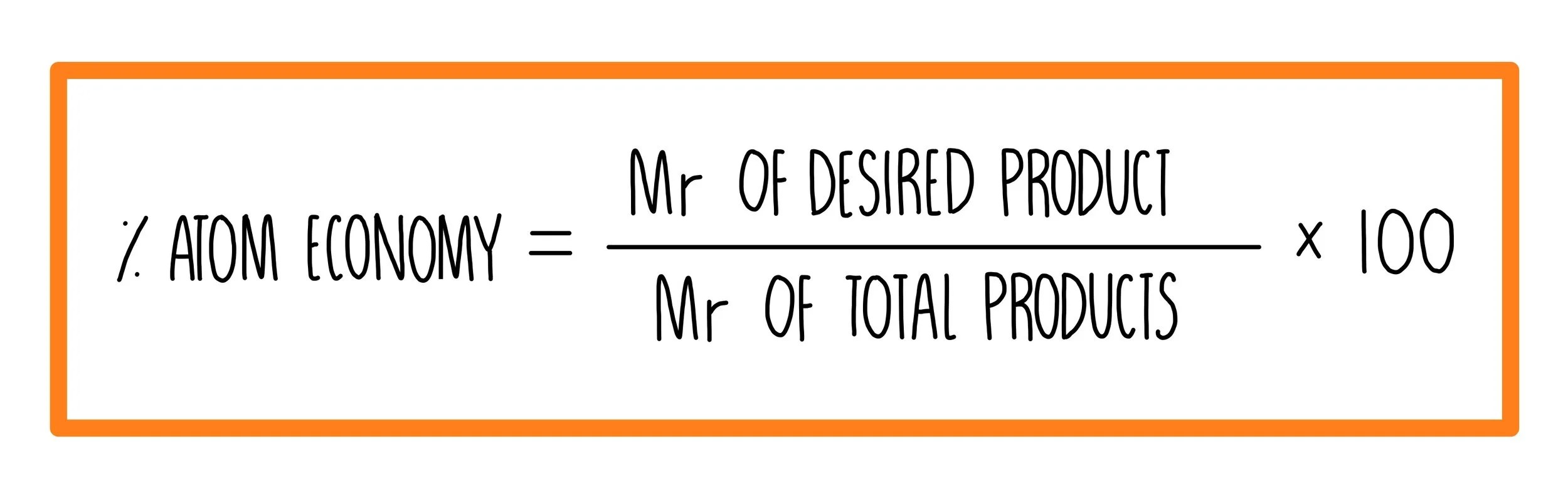
No answers and no good for home learning. Mass of desired useful product. However, with plenty of revision, memorising of the formula's becomes easier, understanding atom economy becomes second nature and applying avagadro's constant will become straight forward. Download all our revision notes as pdfs. No answers and no good for home learning. 22.02.2018 · these worksheets cover a number of topics:. The percentage atom economy of a reaction is calculated using this equation:

No answers and no good for home learning.. 22.02.2018 · these worksheets cover a number of topics: From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Use our chemistry revision … Download all our revision notes as pdfs. The atom economy could also be calculated using mass, instead or mr.. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100

Its sometimes referred to as atom utilisation.. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The percentage atom economy of a reaction is calculated using this equation: In this case, you would divide the mass of the desired product formed by the total mass of all reactants, and then multiply by 100. No answers and no good for home learning. Download all our revision notes as pdfs. 22.02.2018 · these worksheets cover a number of topics: Something went wrong, please try again later. From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. Its sometimes referred to as atom utilisation... Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc.

Use our chemistry revision ….. Atom economy = \(\frac{mass~of~atoms~in~the~desired~product}{total~mass~of~atoms~in~reactants}\) x 100 22.02.2018 · these worksheets cover a number of topics: The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Calculating atom economy atom economy can be calculated using this equation: Its sometimes referred to as atom utilisation. Word and symbol equations, balancing equations, rfm calculations, % yield, % atom economy, % composition of element in a compound, empirical formula, solution calculations etc. From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult.. Its sometimes referred to as atom utilisation.

Yield & atom economy | aqa gcse chemistry | questions & answers. Download all our revision notes as pdfs. From understanding avagadro's contact, to mole calculations, formula's for percentage yield and atom economy, at first this part of the gcse chemistry syllabus seems very difficult. The reaction is as follows: Atom economy = !! x 100 = 4.3% b) calculate the atom economy to form hydrogen by method 2. Its sometimes referred to as atom utilisation.