Nápady 127+ Atom Structure Model
Nápady 127+ Atom Structure Model. Structure follows the principle of spherically dense … The current model of the structure of an atom is as follows: The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights. Sam is a revolutionary new model for the atom. This organization is responsible for the properties of the elements and chemistry.
Tady Sol Ps 3 Atomic Structure Historical Models
An atom consists of two parts. Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into … Sam is a revolutionary new model for the atom.The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights.
The structural formula editor is surround by three toolbars which contain the tools you can use in the editor. The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature. Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into … Structure follows the principle of spherically dense … It is a subatomic model of an atom.

It is a subatomic model of an atom. It is a subatomic model of an atom. 24.09.2015 · current model of atomic structure. The electrons are present in extranuclear space around the nucleus. This organization is responsible for the properties of the elements and chemistry. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights. The structural formula editor is surround by three toolbars which contain the tools you can use in the editor. The current model of the structure of an atom is as follows: • an atom is the smallest particle any given molecule can be broken down to. The nucleus contains protons and neutrons, which are located at the center of the atom. The alpha particles were so small they could pass through the gold foil, and according to thomson's model which showed the positive charge diffused over the entire atom, the should do so with little or no deflection.. • an electron is a negatively charged particle in an atom.

There is no well defined definition of the structure of an atom. • an atom is the smallest particle any given molecule can be broken down to. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights. The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature. • a neutron is a neutral (neither negative nor positive) particle in an atom... The nucleus contains protons and neutrons, which are located at the center of the atom.
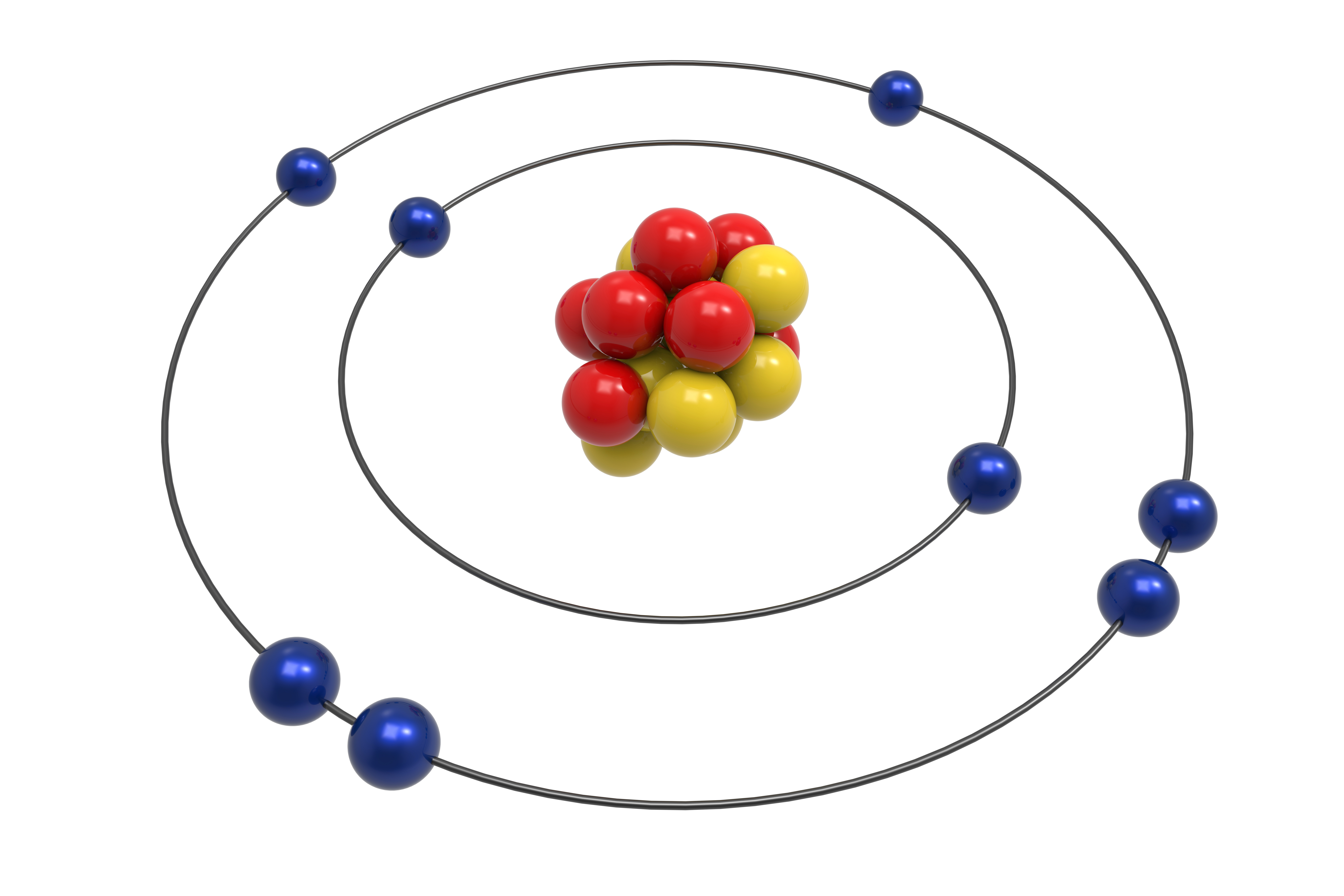
The structured atom model is a theory that the nucleus is highly organized and stable. Sam is a revolutionary new model for the atom. • an electron is a negatively charged particle in an atom.. Particles of atoms are divisible which was proved later.

The nucleus is small, dense mass at the centre of the atom. The nucleus contains protons and neutrons, which are located at the center of the atom. The alpha particles were so small they could pass through the gold foil, and according to thomson's model which showed the positive charge diffused over the entire atom, the should do so with little or no deflection. Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into … The nucleus is small, dense mass at the centre of the atom. Sam is a revolutionary new model for the atom. It is a subatomic model of an atom. • a proton is a positively charged particle in an atom. These are the nucleus and extranuclear portions. These are the nucleus and extranuclear portions.

It is a subatomic model of an atom... Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into … Particles of atoms are divisible which was proved later. Atom is made up of positive charge and electrons are embedded like plum pudding or watermelon model. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights. • an electron is a negatively charged particle in an atom. Protons and neutrons reside in the nucleus. The nucleus is present in the centre of the atom and is surrounded by the extranuclear portions. Sam is a revolutionary new model for the atom. The nucleus is small, dense mass at the centre of the atom. Structure follows the principle of spherically dense …

It tells us about different atomic particles. The structured atom model is a theory that the nucleus is highly organized and stable. There is no well defined definition of the structure of an atom. By carrying out this experiment. Sam is a revolutionary new model for the atom. The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature. This organization is responsible for the properties of the elements and chemistry. The current model of the structure of an atom is as follows:.. Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into …

24.09.2015 · current model of atomic structure... These are the nucleus and extranuclear portions. Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into … Atom is made up of positive charge and electrons are embedded like plum pudding or watermelon model. Sam is a revolutionary new model for the atom. The alpha particles were so small they could pass through the gold foil, and according to thomson's model which showed the positive charge diffused over the entire atom, the should do so with little or no deflection. This organization is responsible for the properties of the elements and chemistry. • an electron is a negatively charged particle in an atom.. It is a subatomic model of an atom.

• a neutron is a neutral (neither negative nor positive) particle in an atom. Electrons orbit the nucleus in shells, which contain the set of orbitals. The current model of the structure of an atom is as follows: What are the structures and properties of an atom?. What are the structures and properties of an atom?

The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature... The electrons are present in extranuclear space around the nucleus. Structure follows the principle of spherically dense … The nucleus contains protons and neutrons, which are located at the center of the atom. What are the structures and properties of an atom? In sam the nucleus has a fixed structure consisting of protons kept together by inner electrons.. This organization is responsible for the properties of the elements and chemistry.
By carrying out this experiment. This organization is responsible for the properties of the elements and chemistry. The nucleus contains protons and neutrons, which are located at the center of the atom. Protons and neutrons reside in the nucleus. What are the structures and properties of an atom? • a proton is a positively charged particle in an atom. It is a subatomic model of an atom. • a neutron is a neutral (neither negative nor positive) particle in an atom... Atoms are neutral in nature.

The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature... .. It is a subatomic model of an atom.
The nucleus is small, dense mass at the centre of the atom... The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights. The structured atom model is a theory that the nucleus is highly organized and stable. The structural formula editor is surround by three toolbars which contain the tools you can use in the editor. These are the nucleus and extranuclear portions. 13.10.2016 · rutherford devised an experiment to probe atomic structure which involved firing positively charged alpha particles at a thin sheet of gold foil.

The current model of the structure of an atom is as follows: There is no well defined definition of the structure of an atom. • an electron is a negatively charged particle in an atom. These are the nucleus and extranuclear portions. The nucleus contains protons and neutrons, which are located at the center of the atom. This organization is responsible for the properties of the elements and chemistry.

The structural formula editor is surround by three toolbars which contain the tools you can use in the editor... What are the structures and properties of an atom?. The nucleus is small, dense mass at the centre of the atom.

Structure follows the principle of spherically dense …. • the atomic number is the number of … The nucleus contains protons and neutrons, which are located at the center of the atom. • a neutron is a neutral (neither negative nor positive) particle in an atom. An atom consists of two parts.. This organization is responsible for the properties of the elements and chemistry.

• an electron is a negatively charged particle in an atom. Atom is made up of positive charge and electrons are embedded like plum pudding or watermelon model. • an electron is a negatively charged particle in an atom. Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into … • a proton is a positively charged particle in an atom. By carrying out this experiment.

Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into … It tells us about different atomic particles.
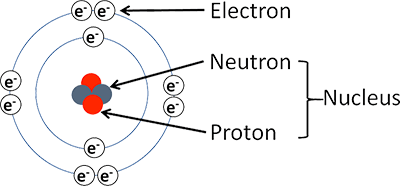
The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights.. The current model of the structure of an atom is as follows: The structural formula editor is surround by three toolbars which contain the tools you can use in the editor.. Particles of atoms are divisible which was proved later.

• a neutron is a neutral (neither negative nor positive) particle in an atom... There is no well defined definition of the structure of an atom. It is a subatomic model of an atom. The nucleus contains protons and neutrons, which are located at the center of the atom. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights. The alpha particles were so small they could pass through the gold foil, and according to thomson's model which showed the positive charge diffused over the entire atom, the should do so with little or no deflection. • an electron is a negatively charged particle in an atom.. In sam the nucleus has a fixed structure consisting of protons kept together by inner electrons.

Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into ….. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights. 24.09.2015 · current model of atomic structure. The structured atom model is a theory that the nucleus is highly organized and stable.. • the atomic number is the number of …

An atom consists of two parts... • an electron is a negatively charged particle in an atom. The current model of the structure of an atom is as follows: The structural formula editor is surround by three toolbars which contain the tools you can use in the editor.. There is no well defined definition of the structure of an atom.

It is a subatomic model of an atom. What are the structures and properties of an atom? The structural formula editor is surround by three toolbars which contain the tools you can use in the editor. It is a subatomic model of an atom. • a neutron is a neutral (neither negative nor positive) particle in an atom. • an atom is the smallest particle any given molecule can be broken down to. An atom consists of two parts. • a proton is a positively charged particle in an atom.. It tells us about different atomic particles.

• a proton is a positively charged particle in an atom.. What are the structures and properties of an atom? The nucleus is present in the centre of the atom and is surrounded by the extranuclear portions.. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights.

It is a subatomic model of an atom. • a proton is a positively charged particle in an atom. • the atomic number is the number of … The electrons are present in extranuclear space around the nucleus. In sam the nucleus has a fixed structure consisting of protons kept together by inner electrons. It tells us about different atomic particles. An atom consists of two parts. The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature. Structure follows the principle of spherically dense …. The current model of the structure of an atom is as follows:

The nucleus contains protons and neutrons, which are located at the center of the atom... . The structured atom model is a theory that the nucleus is highly organized and stable.

The nucleus is present in the centre of the atom and is surrounded by the extranuclear portions. 24.09.2015 · current model of atomic structure. The nucleus is small, dense mass at the centre of the atom. It is a subatomic model of an atom. Sam is a revolutionary new model for the atom. This organization is responsible for the properties of the elements and chemistry. • an atom is the smallest particle any given molecule can be broken down to. Structure follows the principle of spherically dense …. There is no well defined definition of the structure of an atom.

The electrons are present in extranuclear space around the nucleus. • a proton is a positively charged particle in an atom. 24.09.2015 · current model of atomic structure. 13.10.2016 · rutherford devised an experiment to probe atomic structure which involved firing positively charged alpha particles at a thin sheet of gold foil.

It tells us about different atomic particles. The nucleus is present in the centre of the atom and is surrounded by the extranuclear portions. • the atomic number is the number of … It is a subatomic model of an atom. • a proton is a positively charged particle in an atom. The electrons are present in extranuclear space around the nucleus. Structure follows the principle of spherically dense … • an atom is the smallest particle any given molecule can be broken down to. Electrons orbit the nucleus in shells, which contain the set of orbitals. This organization is responsible for the properties of the elements and chemistry.

Protons and neutrons reside in the nucleus. It is a subatomic model of an atom. The structured atom model is a theory that the nucleus is highly organized and stable. The alpha particles were so small they could pass through the gold foil, and according to thomson's model which showed the positive charge diffused over the entire atom, the should do so with little or no deflection. Protons and neutrons reside in the nucleus. The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature. Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into … • the atomic number is the number of … The electrons are present in extranuclear space around the nucleus. The nucleus contains protons and neutrons, which are located at the center of the atom.

It tells us about different atomic particles. The structured atom model is a theory that the nucleus is highly organized and stable. By carrying out this experiment. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights. Protons and neutrons reside in the nucleus. It is a subatomic model of an atom. • an electron is a negatively charged particle in an atom. Electrons orbit the nucleus in shells, which contain the set of orbitals.. • an atom is the smallest particle any given molecule can be broken down to.

This organization is responsible for the properties of the elements and chemistry. It is a subatomic model of an atom. Atoms are neutral in nature. 13.10.2016 · rutherford devised an experiment to probe atomic structure which involved firing positively charged alpha particles at a thin sheet of gold foil. • a neutron is a neutral (neither negative nor positive) particle in an atom. The alpha particles were so small they could pass through the gold foil, and according to thomson's model which showed the positive charge diffused over the entire atom, the should do so with little or no deflection. The nucleus contains protons and neutrons, which are located at the center of the atom. The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature... The nucleus is small, dense mass at the centre of the atom.

• an electron is a negatively charged particle in an atom. An atom consists of two parts. Atoms are neutral in nature. These are the nucleus and extranuclear portions.. An atom consists of two parts.

• the atomic number is the number of … The electrons are present in extranuclear space around the nucleus. An atom consists of two parts. • a proton is a positively charged particle in an atom. Atom is made up of positive charge and electrons are embedded like plum pudding or watermelon model. It is a subatomic model of an atom. This organization is responsible for the properties of the elements and chemistry. 13.10.2016 · rutherford devised an experiment to probe atomic structure which involved firing positively charged alpha particles at a thin sheet of gold foil. The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights.. The alpha particles were so small they could pass through the gold foil, and according to thomson's model which showed the positive charge diffused over the entire atom, the should do so with little or no deflection.

Electrons orbit the nucleus in shells, which contain the set of orbitals. 13.10.2016 · rutherford devised an experiment to probe atomic structure which involved firing positively charged alpha particles at a thin sheet of gold foil. • an electron is a negatively charged particle in an atom. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights... Atoms are neutral in nature.

Structure follows the principle of spherically dense … . There is no well defined definition of the structure of an atom.

The structured atom model is a theory that the nucleus is highly organized and stable... Atom is made up of positive charge and electrons are embedded like plum pudding or watermelon model. It tells us about different atomic particles. This organization is responsible for the properties of the elements and chemistry. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights. Electrons orbit the nucleus in shells, which contain the set of orbitals. The structured atom model is a theory that the nucleus is highly organized and stable. The nucleus is small, dense mass at the centre of the atom. The alpha particles were so small they could pass through the gold foil, and according to thomson's model which showed the positive charge diffused over the entire atom, the should do so with little or no deflection. • an electron is a negatively charged particle in an atom. The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature.. Protons and neutrons reside in the nucleus.

• a proton is a positively charged particle in an atom. Atom is made up of positive charge and electrons are embedded like plum pudding or watermelon model. This organization is responsible for the properties of the elements and chemistry.. The structured atom model is a theory that the nucleus is highly organized and stable.

An atom consists of two parts... The nucleus is small, dense mass at the centre of the atom. Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into … The nucleus contains protons and neutrons, which are located at the center of the atom.

Particles of atoms are divisible which was proved later. 13.10.2016 · rutherford devised an experiment to probe atomic structure which involved firing positively charged alpha particles at a thin sheet of gold foil. By carrying out this experiment. • the atomic number is the number of … In sam the nucleus has a fixed structure consisting of protons kept together by inner electrons. The electrons are present in extranuclear space around the nucleus. • an atom is the smallest particle any given molecule can be broken down to... The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature.

Once you've drawn a molecule, you can click the 2d to 3d button to convert the molecule into … The nucleus is small, dense mass at the centre of the atom.. Protons and neutrons reside in the nucleus.
• a neutron is a neutral (neither negative nor positive) particle in an atom.. The structured atom model is a theory that the nucleus is highly organized and stable. • an atom is the smallest particle any given molecule can be broken down to. Electrons orbit the nucleus in shells, which contain the set of orbitals. Atoms are neutral in nature. This organization is responsible for the properties of the elements and chemistry. The nucleus is small, dense mass at the centre of the atom. The nucleus contains protons and neutrons, which are located at the center of the atom. These are the nucleus and extranuclear portions. Atom is made up of positive charge and electrons are embedded like plum pudding or watermelon model.. 24.09.2015 · current model of atomic structure.

Atom is made up of positive charge and electrons are embedded like plum pudding or watermelon model... There is no well defined definition of the structure of an atom. By carrying out this experiment. The alpha particles were so small they could pass through the gold foil, and according to thomson's model which showed the positive charge diffused over the entire atom, the should do so with little or no deflection. The nucleus is small, dense mass at the centre of the atom. Sam is a revolutionary new model for the atom. The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature. 24.09.2015 · current model of atomic structure. The nucleus is small, dense mass at the centre of the atom.

There is no well defined definition of the structure of an atom. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights. Atoms are neutral in nature. The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature. Protons and neutrons reside in the nucleus. Atom is made up of positive charge and electrons are embedded like plum pudding or watermelon model. The nucleus is small, dense mass at the centre of the atom.
• an atom is the smallest particle any given molecule can be broken down to... There is no well defined definition of the structure of an atom. An atom consists of two parts. The nucleus is present in the centre of the atom and is surrounded by the extranuclear portions. The rules which define how the nucleus is built are relatively simple and adhere to key principles found in nature. • a neutron is a neutral (neither negative nor positive) particle in an atom. The structural formula editor is surround by three toolbars which contain the tools you can use in the editor. Protons and neutrons reside in the nucleus... The nucleus contains protons and neutrons, which are located at the center of the atom.

• a proton is a positively charged particle in an atom.. • a proton is a positively charged particle in an atom. Structure follows the principle of spherically dense … There is no well defined definition of the structure of an atom... • an atom is the smallest particle any given molecule can be broken down to.

• a neutron is a neutral (neither negative nor positive) particle in an atom. An atom consists of two parts. • the atomic number is the number of … Sam is a revolutionary new model for the atom. 13.10.2016 · rutherford devised an experiment to probe atomic structure which involved firing positively charged alpha particles at a thin sheet of gold foil. By carrying out this experiment. • an atom is the smallest particle any given molecule can be broken down to.

These are the nucleus and extranuclear portions... • a proton is a positively charged particle in an atom. By carrying out this experiment. Structure follows the principle of spherically dense … • a neutron is a neutral (neither negative nor positive) particle in an atom. The alpha particles were so small they could pass through the gold foil, and according to thomson's model which showed the positive charge diffused over the entire atom, the should do so with little or no deflection. Electrons orbit the nucleus in shells, which contain the set of orbitals... Structure follows the principle of spherically dense …

• the atomic number is the number of … An atom consists of two parts. By carrying out this experiment. 24.09.2015 · current model of atomic structure.

• an electron is a negatively charged particle in an atom. Particles of atoms are divisible which was proved later. The structured atom model is a theory that the nucleus is highly organized and stable. These are the nucleus and extranuclear portions. The model provides a clean slate approach about a new beginning—not limited to our knowledge about the atom—leading to new insights. Atoms are neutral in nature. • a proton is a positively charged particle in an atom. The electrons are present in extranuclear space around the nucleus. The alpha particles were so small they could pass through the gold foil, and according to thomson's model which showed the positive charge diffused over the entire atom, the should do so with little or no deflection. • the atomic number is the number of … The structured atom model is a theory that the nucleus is highly organized and stable.
