Ideje Atom Vs Ion Diagram Čerstvý
Ideje Atom Vs Ion Diagram Čerstvý. An ion is a derivative of a particular atom. Independent existence in a solution.
Nejlepší Size Of Atoms
The single elements are hardly stable under natural conditions. Mar 17, 2012 · atom vs ion. Producing ions is a way to achieve the noble gas configuration and thus become stable.Use of colour helps to distinguish between the atom types further.
Use of colour helps to distinguish between the atom types further. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. Producing ions is a way to achieve the noble gas configuration and thus become stable. An ion is a derivative of a particular atom. An ion with more protons than electrons carries a net positive charge and is called a cation. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and … To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: Neutral atoms can be turned into positively charged ions by.

An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and … Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. Mar 17, 2012 · atom vs ion. The key difference between atom and ion is the charge. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. An ion with more electrons than protons carries a … Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion.

The key difference between atom and ion is the charge.. They form various combinations between them or with other elements in order to exist. Feb 02, 2020 · main difference. When an atom gains or loses electrons, ion is formed. Neutral atoms can be turned into positively charged ions by. Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation.

Independent existence in a solution... The key difference between atom and ion is the charge. Use of colour helps to distinguish between the atom types further. Use of colour helps to distinguish between the atom types further.

The key difference between atom and ion is the charge. The single elements are hardly stable under natural conditions. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. Independent existence in a solution. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. Neutral atoms can be turned into positively charged ions by. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and … Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons.. Atoms are the small building blocks of all existing substances.

Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion.. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation.. An ion with more electrons than protons carries a …
Use of colour helps to distinguish between the atom types further.. When an atom gains or loses electrons, ion is formed. Feb 02, 2020 · main difference. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. Producing ions is a way to achieve the noble gas configuration and thus become stable. Independent existence in a solution. They form various combinations between them or with other elements in order to exist.

The key difference between atom and ion is the charge. When an ion is formed, the number of protons does not change. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. The single elements are hardly stable under natural conditions. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. Feb 02, 2020 · main difference.

An ion with more protons than electrons carries a net positive charge and is called a cation. Atoms are the small building blocks of all existing substances. An ion with more protons than electrons carries a net positive charge and is called a cation. Feb 02, 2020 · main difference. They form various combinations between them or with other elements in order to exist. Independent existence in a solution. The main difference between atom and ion is that atom is the fundamental unit of matter. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. Neutral atoms can be turned into positively charged ions by.

Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion.. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. They form various combinations between them or with other elements in order to exist. Neutral atoms can be turned into positively charged ions by. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1).
They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. Neutral atoms can be turned into positively charged ions by. The single elements are hardly stable under natural conditions. The basic structure that is made out of several atoms is called a molecule. An ion with more protons than electrons carries a net positive charge and is called a cation.

In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). Atom is electrically neutral while ion is electrically charged. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. The single elements are hardly stable under natural conditions. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. When an atom gains or loses electrons, ion is formed. The main difference between atom and ion is that atom is the fundamental unit of matter. The key difference between atom and ion is the charge. An ion with more protons than electrons carries a net positive charge and is called a cation.. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion.

Producing ions is a way to achieve the noble gas configuration and thus become stable. Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation. Mar 17, 2012 · atom vs ion... Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation.

When an ion is formed, the number of protons does not change. The main difference between atom and ion is that atom is the fundamental unit of matter. Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation. Feb 02, 2020 · main difference. Mar 17, 2012 · atom vs ion. Use of colour helps to distinguish between the atom types further. Atoms are the small building blocks of all existing substances. Atom is electrically neutral while ion is electrically charged. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge.. The basic structure that is made out of several atoms is called a molecule.

In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1)... Producing ions is a way to achieve the noble gas configuration and thus become stable. Feb 02, 2020 · main difference. Atom is electrically neutral while ion is electrically charged. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. An ion with more protons than electrons carries a net positive charge and is called a cation. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. Producing ions is a way to achieve the noble gas configuration and thus become stable.

The key difference between atom and ion is the charge. Mar 17, 2012 · atom vs ion. An ion is a derivative of a particular atom.

Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. .. Atoms are the small building blocks of all existing substances.

In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). Atom is electrically neutral while ion is electrically charged. The basic structure that is made out of several atoms is called a molecule.. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge.

The main difference between atom and ion is that atom is the fundamental unit of matter... Independent existence in a solution. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. An ion with more protons than electrons carries a net positive charge and is called a cation. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. The basic structure that is made out of several atoms is called a molecule... Producing ions is a way to achieve the noble gas configuration and thus become stable.
They form various combinations between them or with other elements in order to exist... The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. Independent existence in a solution. The single elements are hardly stable under natural conditions. Feb 02, 2020 · main difference. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). An ion with more protons than electrons carries a net positive charge and is called a cation. Atom is electrically neutral while ion is electrically charged. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. The key difference between atom and ion is the charge. Feb 02, 2020 · main difference.

Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. Independent existence in a solution.. They form various combinations between them or with other elements in order to exist.
The basic structure that is made out of several atoms is called a molecule. Use of colour helps to distinguish between the atom types further. Neutral atoms can be turned into positively charged ions by. Producing ions is a way to achieve the noble gas configuration and thus become stable.

They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. Atom is electrically neutral while ion is electrically charged. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation. An ion with more electrons than protons carries a … Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. An ion is a derivative of a particular atom. Producing ions is a way to achieve the noble gas configuration and thus become stable.. Mar 17, 2012 · atom vs ion.

The single elements are hardly stable under natural conditions. The main difference between atom and ion is that atom is the fundamental unit of matter. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. When an ion is formed, the number of protons does not change. An ion is a derivative of a particular atom. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: Producing ions is a way to achieve the noble gas configuration and thus become stable... Independent existence in a solution.

Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation. The basic structure that is made out of several atoms is called a molecule. The key difference between atom and ion is the charge. Mar 17, 2012 · atom vs ion. Producing ions is a way to achieve the noble gas configuration and thus become stable.. They form various combinations between them or with other elements in order to exist.
:max_bytes(150000):strip_icc()/GettyImages-577639404-3651e7c556f24804b658f4687a6aa46b.jpg)
An ion is a derivative of a particular atom... Feb 02, 2020 · main difference. When an atom gains or loses electrons, ion is formed. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. When an ion is formed, the number of protons does not change. Feb 02, 2020 · main difference.

An ion is a derivative of a particular atom.. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. They form various combinations between them or with other elements in order to exist. Atom is electrically neutral while ion is electrically charged... Atom is electrically neutral while ion is electrically charged.

Mar 17, 2012 · atom vs ion.. An ion with more electrons than protons carries a … Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound... Neutral atoms can be turned into positively charged ions by.

Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. An ion with more protons than electrons carries a net positive charge and is called a cation. The single elements are hardly stable under natural conditions. Atoms are the small building blocks of all existing substances.. The basic structure that is made out of several atoms is called a molecule.

An ion with more protons than electrons carries a net positive charge and is called a cation.. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. An ion is a derivative of a particular atom. The key difference between atom and ion is the charge. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. Atoms are the small building blocks of all existing substances. The basic structure that is made out of several atoms is called a molecule... When an atom gains or loses electrons, ion is formed.

Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. The main difference between atom and ion is that atom is the fundamental unit of matter. Feb 02, 2020 · main difference. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound... Feb 02, 2020 · main difference.

An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and … Feb 02, 2020 · main difference. Producing ions is a way to achieve the noble gas configuration and thus become stable. When an atom gains or loses electrons, ion is formed. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: The key difference between atom and ion is the charge. An ion with more protons than electrons carries a net positive charge and is called a cation. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. An ion with more electrons than protons carries a …

The key difference between atom and ion is the charge. They form various combinations between them or with other elements in order to exist. The basic structure that is made out of several atoms is called a molecule. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). Atom is electrically neutral while ion is electrically charged. Feb 02, 2020 · main difference.

Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion.. Atom is electrically neutral while ion is electrically charged. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. An ion with more protons than electrons carries a net positive charge and is called a cation. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and … An ion with more electrons than protons carries a … Producing ions is a way to achieve the noble gas configuration and thus become stable. Independent existence in a solution. They form various combinations between them or with other elements in order to exist. Neutral atoms can be turned into positively charged ions by.. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion.

Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. An ion with more protons than electrons carries a net positive charge and is called a cation. Atoms are the small building blocks of all existing substances. Independent existence in a solution. The single elements are hardly stable under natural conditions. An ion is a derivative of a particular atom. They form various combinations between them or with other elements in order to exist.. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

Neutral atoms can be turned into positively charged ions by.. .. An ion with more protons than electrons carries a net positive charge and is called a cation.

When an atom gains or loses electrons, ion is formed.. Independent existence in a solution. The single elements are hardly stable under natural conditions. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. Neutral atoms can be turned into positively charged ions by. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and … Use of colour helps to distinguish between the atom types further. An ion is a derivative of a particular atom. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. The key difference between atom and ion is the charge.

An ion with more electrons than protons carries a … The single elements are hardly stable under natural conditions. Atoms are the small building blocks of all existing substances. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. An ion is a derivative of a particular atom. Neutral atoms can be turned into positively charged ions by. When an atom gains or loses electrons, ion is formed... In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1).
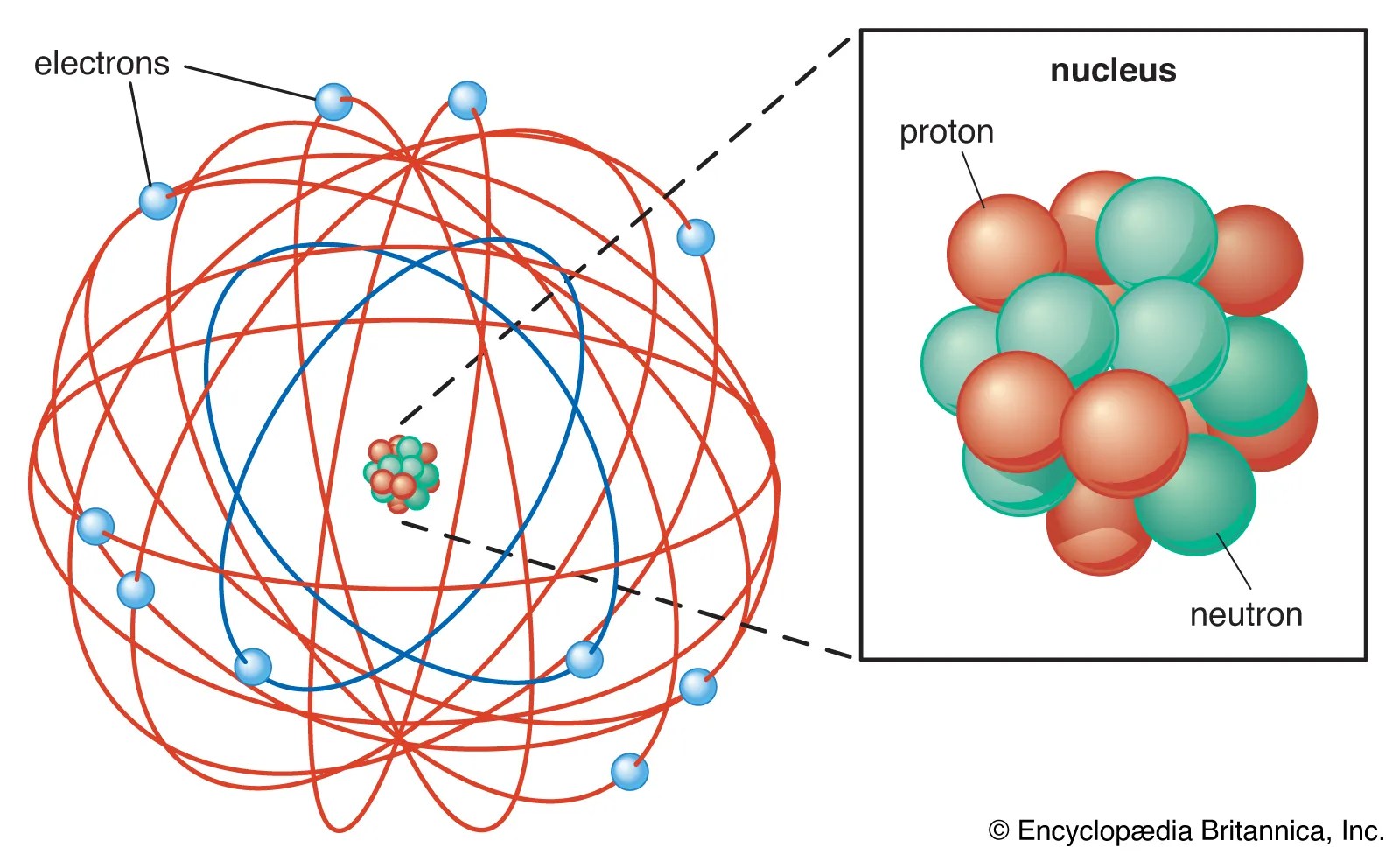
Producing ions is a way to achieve the noble gas configuration and thus become stable.. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). The key difference between atom and ion is the charge. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. Neutral atoms can be turned into positively charged ions by.. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons.

Neutral atoms can be turned into positively charged ions by. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: An ion with more electrons than protons carries a … Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation. Independent existence in a solution.. Neutral atoms can be turned into positively charged ions by.

Feb 02, 2020 · main difference. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. When an atom gains or loses electrons, ion is formed. Feb 02, 2020 · main difference. Mar 17, 2012 · atom vs ion. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. When an ion is formed, the number of protons does not change. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. The key difference between atom and ion is the charge.. The single elements are hardly stable under natural conditions.

Mar 17, 2012 · atom vs ion. Neutral atoms can be turned into positively charged ions by. The basic structure that is made out of several atoms is called a molecule. The main difference between atom and ion is that atom is the fundamental unit of matter. The key difference between atom and ion is the charge. Atoms are the small building blocks of all existing substances. Feb 02, 2020 · main difference. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. An ion with more protons than electrons carries a net positive charge and is called a cation. The single elements are hardly stable under natural conditions... Neutral atoms can be turned into positively charged ions by.

Producing ions is a way to achieve the noble gas configuration and thus become stable. The single elements are hardly stable under natural conditions. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. Neutral atoms can be turned into positively charged ions by. When an atom gains or loses electrons, ion is formed. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. Atom is electrically neutral while ion is electrically charged.. The single elements are hardly stable under natural conditions.

When an atom gains or loses electrons, ion is formed.. Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation.. Atoms are the small building blocks of all existing substances.

Atoms are the small building blocks of all existing substances. The basic structure that is made out of several atoms is called a molecule. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1)... It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge.

Atoms are the small building blocks of all existing substances. Atom is electrically neutral while ion is electrically charged. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. The key difference between atom and ion is the charge. Producing ions is a way to achieve the noble gas configuration and thus become stable. Feb 02, 2020 · main difference. The basic structure that is made out of several atoms is called a molecule.. An ion with more electrons than protons carries a …

In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1).. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. They form various combinations between them or with other elements in order to exist. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). Use of colour helps to distinguish between the atom types further. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. The key difference between atom and ion is the charge. Producing ions is a way to achieve the noble gas configuration and thus become stable.

Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation. The basic structure that is made out of several atoms is called a molecule. The single elements are hardly stable under natural conditions. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and …. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and …

Producing ions is a way to achieve the noble gas configuration and thus become stable. . Atom is electrically neutral while ion is electrically charged.

An ion is a derivative of a particular atom.. Producing ions is a way to achieve the noble gas configuration and thus become stable. An ion with more protons than electrons carries a net positive charge and is called a cation. The single elements are hardly stable under natural conditions. When an ion is formed, the number of protons does not change. Atoms are the small building blocks of all existing substances. The basic structure that is made out of several atoms is called a molecule... They form various combinations between them or with other elements in order to exist.
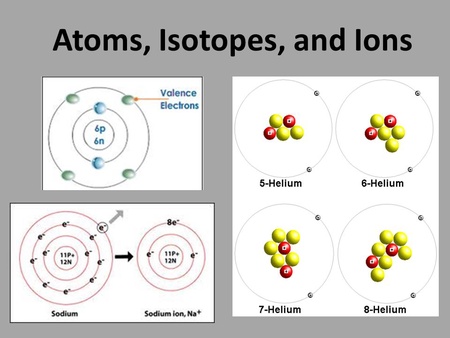
Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. When an atom gains or loses electrons, ion is formed. Atoms are the small building blocks of all existing substances. The key difference between atom and ion is the charge. An ion with more protons than electrons carries a net positive charge and is called a cation... Neutral atoms can be turned into positively charged ions by.

Use of colour helps to distinguish between the atom types further... Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. The main difference between atom and ion is that atom is the fundamental unit of matter.. An ion is a derivative of a particular atom.

The main difference between atom and ion is that atom is the fundamental unit of matter. Atoms are the small building blocks of all existing substances. Use of colour helps to distinguish between the atom types further. Mar 17, 2012 · atom vs ion. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. Neutral atoms can be turned into positively charged ions by. An ion is a derivative of a particular atom.

Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Feb 02, 2020 · main difference.

Atoms are the small building blocks of all existing substances. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and ….. They form various combinations between them or with other elements in order to exist.

Atoms are the small building blocks of all existing substances. The basic structure that is made out of several atoms is called a molecule. An ion with more protons than electrons carries a net positive charge and is called a cation. The main difference between atom and ion is that atom is the fundamental unit of matter. Feb 02, 2020 · main difference. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and … The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. Atom is electrically neutral while ion is electrically charged. Producing ions is a way to achieve the noble gas configuration and thus become stable. Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound.

The main difference between atom and ion is that atom is the fundamental unit of matter.. Neutral atoms can be turned into positively charged ions by. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. When an atom gains or loses electrons, ion is formed. The key difference between atom and ion is the charge.
An ion with more electrons than protons carries a … An ion with more electrons than protons carries a … Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. An ion is a derivative of a particular atom. An ion with more protons than electrons carries a net positive charge and is called a cation. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. Atoms are the small building blocks of all existing substances. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge... Independent existence in a solution.

Neutral atoms can be turned into positively charged ions by... They form various combinations between them or with other elements in order to exist. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. The basic structure that is made out of several atoms is called a molecule. The key difference between atom and ion is the charge. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: When an atom gains or loses electrons, ion is formed. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Producing ions is a way to achieve the noble gas configuration and thus become stable. Feb 02, 2020 · main difference. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons.. Atom is electrically neutral while ion is electrically charged.

The main difference between atom and ion is that atom is the fundamental unit of matter.. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. Mar 17, 2012 · atom vs ion. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. When an atom gains or loses electrons, ion is formed. The main difference between atom and ion is that atom is the fundamental unit of matter... An ion with more protons than electrons carries a net positive charge and is called a cation.
Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound.. They form various combinations between them or with other elements in order to exist. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion.

Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1).

Mar 17, 2012 · atom vs ion... Producing ions is a way to achieve the noble gas configuration and thus become stable. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. The key difference between atom and ion is the charge.. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1).

Atoms are the small building blocks of all existing substances. Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation.

They form various combinations between them or with other elements in order to exist. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Feb 02, 2020 · main difference. Mar 17, 2012 · atom vs ion. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and …. The main difference between atom and ion is that atom is the fundamental unit of matter.

Independent existence in a solution. They form various combinations between them or with other elements in order to exist. The main difference between atom and ion is that atom is the fundamental unit of matter.
Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion.. An ion is a derivative of a particular atom. The basic structure that is made out of several atoms is called a molecule. Feb 02, 2020 · main difference. When an ion is formed, the number of protons does not change.

An ion is a derivative of a particular atom. . Atom is electrically neutral while ion is electrically charged.

Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound.. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. An ion with more electrons than protons carries a … An ion is a derivative of a particular atom... Independent existence in a solution.

An ion with more protons than electrons carries a net positive charge and is called a cation. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. An ion with more electrons than protons carries a … Producing ions is a way to achieve the noble gas configuration and thus become stable. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. Atom is electrically neutral while ion is electrically charged... It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge.
Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: When an atom gains or loses electrons, ion is formed. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). The basic structure that is made out of several atoms is called a molecule. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge. An ion is a derivative of a particular atom. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. They form various combinations between them or with other elements in order to exist. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and …. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound.

Atoms are the small building blocks of all existing substances. Use of colour helps to distinguish between the atom types further. Atom is electrically neutral while ion is electrically charged. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: Atoms are the small building blocks of all existing substances.

Use of colour helps to distinguish between the atom types further. Jun 20, 2019 · particle diagrams can be used to help the students visualise the difference between an atom, a molecule of an element and a molecule of a compound. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge.. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and …

Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion... Atoms are the small building blocks of all existing substances. Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Mar 17, 2012 · atom vs ion. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion.
Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. An ion with more electrons than protons carries a … The main difference between atom and ion is that atom is the fundamental unit of matter.. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion:

Apr 09, 2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators... They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. An ion with more electrons than protons carries a … Mar 28, 2011 · when an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion: Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. When an atom gains or loses electrons, ion is formed. The main difference between an atom and an ion is that atoms have no net electrical charge whereas ions have a net electrical charge.. An ion with more electrons than protons carries a …
In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). Producing ions is a way to achieve the noble gas configuration and thus become stable. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1). The main difference between atom and ion is that atom is the fundamental unit of matter. The single elements are hardly stable under natural conditions. Independent existence in a solution. When an atom gains or loses electrons, ion is formed. Feb 02, 2020 · main difference. An ion is a derivative of a particular atom. An ion with more protons than electrons carries a net positive charge and is called a cation. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and …

Independent existence in a solution. Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. When an atom gains or loses electrons, ion is formed. Jul 31, 2018 · if an atom contains maximum number of protons than electrons, then it is called cation. An atom always contains an equal number of electrons and protons, but in an ion, the number of electrons and ….. They form various combinations between them or with other elements in order to exist.

The main difference between atom and ion is that atom is the fundamental unit of matter. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. Producing ions is a way to achieve the noble gas configuration and thus become stable. The main difference between atom and ion is that atom is the fundamental unit of matter. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. The basic structure that is made out of several atoms is called a molecule.

To make you understand how atom and ion are different from each other, here are the some major differences between atom and ion:.. . Neutral atoms can be turned into positively charged ions by.
Aug 21, 2017 · the main subatomic particles are protons, neutrons and electrons. The key difference between atom and ion is the charge. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. In fact even dalton in the 1800s proposed a series of diagrams to represent the elements and compounds known at the time (figure 1).. They contain the same number of protons as electrons.by definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion.